|
To enable end-to-end transparency for all parties, timely availability of standardized and digitalized features is paramount. This lack of timely availability of data is a key obstacle in the implementation of QbD, as it requires real-time analytics to facilitate informed design optimization. 
The only incentive for study specialists to transcribe content to systems are management reporting needs. Additionally, the Tufts Center for the Study of Drug Development (CSDD) found that in 2014-2018, it took an average of 145 days for the study startup period, up 13.7 percent from the prior 2008-2013 period. This is supported by the current industry standard for TMF inspection readiness, which is within 30 days of site activation milestone, with 52 percent (131 artifacts of the 250 Trial Master File (TMF) Reference Model V3.2.1) all still being electronic. 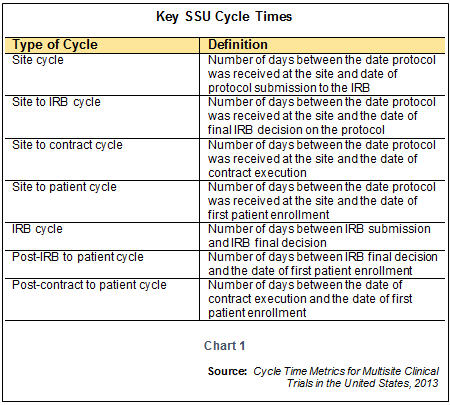
Data are first transcribed into systems, such as study startup or clinical trial management systems, weeks or even months after being captured. Study startup content is sourced over 90 percent electronically from Word, PDF, and Excel documents rather than being captured digitally in databases. With QbD regulatory awareness, the onus is on the industry to implement changes that can reduce the cost and time to bring medical products to market.Įnable and Require Proactive Study Design. These can all be mitigated by study design optimization and review prior to the first protocol approval. Second, it uses principles to design the simplest and most cost-effective solution to mitigate risks.Įxamples of such early identifiable study risks are a need for novel or local Institutional Review Board (IRB) sites, IRB study disapproval from poor study quality, study participant underenrollment, and uncoordinated planning causing delays and unexpected costs. First, it provides an approach to identify risks many months before a protocol is approved. In addition, 10.9 percent of initiated sites are never activated - a constant figure over the past 20 years, according to Tufts - with sites being managed by CROs being activated more often.To overcome this challenge, the International Council for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) issued E8(R1), General Considerations for Clinical Studies, introducing quality by design (QbD).Ĭan QbD be the panacea to untangle the current quality testing-based GCP? Yes, for two reasons. More than half of sponsors still rely on spreadsheets, compared to just under one-third of CROs. This was “very telling because there isn’t a lot of research out there comparing cycle times between sponsors and CROs,” Lamberti said.ĬROs make more use of advanced technology solutions for the study initiation process than drug sponsors, according to Tufts researchers, with 47 percent of CROs saying they used clinical trial management systems compared to only 28 percent of sponsors. Tufts researchers found that CROs are more efficient than drug sponsors, completing initiation an average of 5.6 weeks faster for repeat sites and 11 weeks faster for first-time sites. Sharing information with sites early on to assess study feasibility could enhance the site selection process, according to the Tufts report. 
“It’s happening, but it’s just not happening fast enough,” she said.Īnother potential solution, Lamberti said, is pooling data from clinical sites to counter the siloing of data common among drug manufacturers. 
Sponsors are investing in technology and working to make contracting and budgeting negotiations - which can be a significant drag on the process - more efficient, Lamberti told CWWeekly. Tufts CSDD senior research fellow Mary Jo Lamberti, who led the analysis, said drugmakers are trying to improve the timeliness of the site initiation process. Nearly three in 10 sites were new, with no prior history of working with a sponsor or CRO. Tufts researchers found the widest variations among respondents in site identification cycle times, indicating “highly inconsistent practices,” the report said. Respondents reported that 61 percent of total cycle time was associated with study start-up activities such as contract and budget negotiations. The process of selecting clinical trial sites and launching studies, beginning with site identification and ending with study start-up completion, takes an average of 31.4 weeks for Phase II and III studies - a full month longer than the average seen 10 years ago, according to a survey conducted by the Tufts Center for the Study of Drug Development.īetween 30 and 40 percent of 590 sponsors and CROs said they were somewhat or completely unsatisfied with their processes for site initiation, according to the study.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
